Introduction¶
In this tutorial, we will go through the steps of building spatial RNA graphs for “unsupervised” spatial neighbourbood learning using graph neural network models. Here we focus on spatial transcriptomics data from imaging-based platforms which produce the list of detected transcripts with physical coordinates in the tissue space.
For the purpose of demonstration, we will analyse a synthetic dataset generated using the simulation module from ficture. Briefly, transcripts from ten distinct cell types were simulated and arranged in three shapes in the 2D space. Two cell types have been placed randomly (scattered) across the space, and the remaining cells have been more restricted to a particular area. For more information regarding the simulation data please refer to tutorial 01. For downloading a copy of the data used in this tutorial, it is available on Zenoto.
import pandas as pd
import numpy as np
import matplotlib.pyplot as plt
from torch_geometric.nn import radius_graph
from torch_geometric import seed_everything
import torch
import os.path as osp
import time
import torch.nn.functional as F
from torch_geometric.loader import LinkNeighborLoader,NeighborLoader
from torch_geometric.nn import GraphSAGE
from torch_geometric.data import Data
import random
from pathlib import Path
from torch_geometric.loader import DataLoader
## for reproduciable results
seed = 1024
random.seed(seed) # python random seed
np.random.seed(seed) # numpy random seed
torch.manual_seed(seed) # pytorch random seed
torch.backends.cudnn.deterministic = True
torch.backends.cudnn.benchmark = False
seed_everything(seed)
Apply GNN to learn spatial neighbourhoods of molecules in the tissue¶
Building RNA-transcript spatial graphs¶
We next construct a spatial graph for all the mRNA transcripts in the simulated dataset. In this graph, each node represents a transcript, we connect nodes when their physical distance is smaller than a radius_r, i.e., edges are added between transcripts that sit close (< radius_r)in the physical space. We initialize each node’s input feature with the transcript’s gene labels after one-hot-encoding transformation. The graph is stored as torch_geometric.data.Data.
Required directory structure¶
We will use the SpatialRNA function for constructing the graph data from the input transcripts. SpatialRNA expects input files in a certain directory structure, for example, for sample_x,
./data_dir/sample_x/raw/sample_x.csv,where sample_x.csv is the CSV file with list of detected transcripts (aftering removing non-gene transcripts, i.e., control probes).
Now run SpatialRNA for constructing the graph
For this simulated dataset, we have transcripts originating from 500 genes. It means the initial input feature vector for each transcript/node in the graph will be a vector of length 500.
gene_list = np.unique(pd.read_csv("../data/feature.tsv.gz",sep="\t").gene.values) ## find and substitute this with your gene_panel information for your real data
gene_list[:10] # show first 10 genes
array(['2310065F04Rik', '2610528A11Rik', '4931406C07Rik', 'AA467197',
'Abcb11', 'Acaa1b', 'Acsm2', 'Acta1', 'Actc1', 'Actn2'],
dtype=object)x = torch.tensor(np.arange(gene_list.shape[0]))
one_hot_encoding = dict(zip(gene_list, F.one_hot(x, num_classes=gene_list.shape[0])))
#one_hot_encoding["Abcb11"] # show one-hot encoding for gene Abcb11
tensor([0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0])It means transcripts with gene label Abcb11 are encoded with the vector shown above. However, for reducing storage and I/O we simply store the features of the nodes using integers, and we will convert integer numbers to one-hot-encoding vectors during model training and inference steps.
gene_int = dict(zip(gene_list, torch.tensor(np.arange(gene_list.shape[0]))))
gene_int["Abcb11"] # show integer encoding for gene Abcb11
tensor(4)We next will use the x,y locations of the transcripts to build a radius-based graph using the SpatialRNA dataset constructor. The value of radius is essential, which determins the size of spatial aggregation (spatial smoothing). We use a radius of 3.0 for this tutorial. The simulation data is managable without tiling, for illustration we set the number of tiles to 2.
ls ../data/sim_sample/raw/
sim_sample.csv
from spatialrna import SpatialRNA
sample_name = "sim_sample"
dataset_dir = Path("../data/")
# Create the SpatialRNA object with the specified parameters
data = SpatialRNA(
root = dataset_dir / f'{sample_name}/' ,
sample_name=sample_name,
one_hot_encoding=gene_int,
num_tiles = 2,
dim_x = "X", # x coordinate column name in the CSV file
dim_y = "Y", # y coordinate column name in the CSV file
tile_by_dim="Y", # tile by dimension, can be "X" or "Y"
process_mode="tile", # create tile graph, currently only one tile.
load_type="tile", # load_type can be "blank", no data will be loaded; "graph", load tile graph; "subgraph", load the sampled subgraph.
feature_col="gene", # feature column name in the CSV file
force_reload=True,
process_tile_ids=[0,1], ## Recursivly process first and the second tile. Can choose to only process one tile per process, and use slurm batch job to batch process.
num_neighbours=[-1,-1], # number of neighbours for each layer, -1 means all neighbours
radius_r=3.0 #
)
Processing...
Processing raw file ../data/sim_sample/raw/sim_sample.csv
To create processed files ['../data/sim_sample/processed/sim_sample_data_tile0.pt', '../data/sim_sample/processed/sim_sample_data_tile1.pt']
Raw data shape 557521
tile core area shape (277967, 8)
left padding shape 0
right padding shape 6046
core area plus paddings, shape (284013, 9)
tile core area shape (279554, 8)
left padding shape 7428
right padding shape 0
core area plus paddings, shape (286982, 9)
loading from file ../data/sim_sample/processed/sim_sample_data_tile0.pt
Done!
We can inspect the generate radius graph data:
data = data[0]
data
Data(x=[284013], edge_index=[2, 23324208], trans_id=[284013], core_mask=[284013])Data files were created in the processed dir
ls ../data/sim_sample/processed/We now constructed the data object for GNN training, which consists of
x: contains the initial feature matrix for nodes in the graph;edge_index: contains the list of edges in the format of a pair of nodes.
In an ‘unsupervised’-training setting, true labels are not known and not used for training. In the case of training a GNN model for label (such as cell type) prediction, please check the advanced use cases.
data.x,data.edge_index
(tensor([ 54, 109, 293, ..., 479, 374, 479]),
tensor([[ 81, 302, 170, ..., 283976, 283980, 283990],
[ 0, 0, 0, ..., 284012, 284012, 284012]]))Construct data loader¶
In an ‘unsupervise’ training setting, model training is achieved by solving the edges (link) prediction task. Firstly, we build a training data loader which loads the specified number of graphs as one data batch. From the data batch, we further generate/sample mini-batches of graphs.
We first construct a onDiskDataLoader class that essentially records our generated .pt (graph object) files for our input samples tiles.
import spatialrna.spatialrna_ondisk as spod
## if you had prepared subgraphs.pt
## in ../data/samplename/subgraph/samplename_subgraph_data_tile0.pt and would like to train over the subgraph,
## you can change the pt_dir to "subgraph"
myod = spod.SpatialRNAOnDiskDataset(root = "../data/",pt_dir="processed")
myod.len()
Processing...
Done!
2To inspect the graph data in myod, you can use myod.get, see more at link
## get the first
myod.get(0)
Data(x=[284013], edge_index=[2, 23324208], trans_id=[284013], core_mask=[284013])# Uncomment if splitting the graph tiles in myod to Train and Validation.
# Split indices: 80% train, 20% validation
#
# indices = list(range(len(myod)))
# train_idx, val_idx = train_test_split(indices, test_size=0.2, random_state=42)
# train_idx[0:5], val_idx[0:5],len(train_idx),
# use the two tiles for training
train_idx = [0,1]
train_dataset = myod.index_select(train_idx)
train_loader = DataLoader(train_dataset, batch_size=2, shuffle=True, num_workers=2)
#val_loader = DataLoader(val_dataset, batch_size=20, shuffle=False, num_workers=2)
Construct GNN model, GraphSAGE¶
Now we have prepared our DataLoader object. Next we construct a 2-hop GraphSAGE model with hidden channel size 50.
device = torch.device('cuda' if torch.cuda.is_available() else 'cpu')
number_class = 500
model = GraphSAGE(
number_class,
hidden_channels=50,
num_layers=2).to(device)
torch.cuda.is_available()
TrueTrain the GraphSAGE model via link prediction¶
## use the train function
import spatialrna.train_val as train_val
Training loss is constructed by predicting whether a pair of nodes should be linked (with an edge) based on the current latent representations produced by the model.
It is not necessary to train the model using all edgegs in the all the tile graphs, and we can sample a certain number of edges for training. In the code chunk below, we sample 50k edges per data batch (batch_size = 2 tiles as defined above) and assign them the positive labels (1). Correspondingly, we randomly construct the same number of negative edges by joining the starting nodes with a randomly sampled node in the data.
We specify the number of neighbours to sample for a seed node as [20, 10], which means 20 and 10 neighbours are sampled respectively at the first and the second hop in its neighbourhood.
### Run the training and testing process for 20 epochs
optimizer = torch.optim.Adam(model.parameters(), lr=0.001)
from time import time
times = []
for epoch in range(1, 21):
start = time()
loss,acc = train_val.train(
model=model,
train_loader=train_loader,
device=device,
num_classes=number_class,
optimizer=optimizer,
num_neighbors=[20,10],
num_train_edges=50000,
verbose=False)
print(f'Epoch: {epoch:03d}, Loss: {loss:.4f}, Acc: {acc:.3f}')
times.append(time() - start)
print(f"Median time per epoch: {torch.tensor(times).median():.4f}s")
Epoch: 001, Loss: 0.6812, Acc: 0.583
Epoch: 002, Loss: 0.5653, Acc: 0.737
Epoch: 003, Loss: 0.4896, Acc: 0.757
Epoch: 004, Loss: 0.4694, Acc: 0.783
Epoch: 005, Loss: 0.4595, Acc: 0.804
Epoch: 006, Loss: 0.4562, Acc: 0.820
Epoch: 007, Loss: 0.4498, Acc: 0.836
Epoch: 008, Loss: 0.4459, Acc: 0.840
Epoch: 009, Loss: 0.4424, Acc: 0.843
Epoch: 010, Loss: 0.4433, Acc: 0.847
Epoch: 011, Loss: 0.4408, Acc: 0.864
Epoch: 012, Loss: 0.4366, Acc: 0.885
Epoch: 013, Loss: 0.4342, Acc: 0.893
Epoch: 014, Loss: 0.4360, Acc: 0.897
Epoch: 015, Loss: 0.4325, Acc: 0.905
Epoch: 016, Loss: 0.4320, Acc: 0.912
Epoch: 017, Loss: 0.4336, Acc: 0.910
Epoch: 018, Loss: 0.4318, Acc: 0.912
Epoch: 019, Loss: 0.4317, Acc: 0.912
Epoch: 020, Loss: 0.4292, Acc: 0.914
Median time per epoch: 8.6095s
Obtain embedding for each transcript for downstream analysis¶
Model parameters have now been trained. We feed the graph data to get the latent embeddings for all transcripts for downstream analysis. We can obtain the embedding for each tile graph using the inference function.
train_val.inference(
model=model,
device=device,
sample_name="sim_sample",
root="../data/sim_sample/",
tile_id=[0,1],
num_classes=number_class,
num_neighbors=[20,10])
# subgraph_loader = NeighborLoader(
# data,
# #input_nodes=data.x,
# #num_neighbors=[-1],
# num_neighbors=[10,10],
# batch_size=1024,
# replace=False,
# shuffle=False,
# subgraph_type = "bidirectional")
/mnt/beegfs/mccarthy/general/backed_up/rlyu/Projects/spatialrna_dev0.2/spatialrna/spatialrna/train_val.py:297: FutureWarning: You are using `torch.load` with `weights_only=False` (the current default value), which uses the default pickle module implicitly. It is possible to construct malicious pickle data which will execute arbitrary code during unpickling (See https://github.com/pytorch/pytorch/blob/main/SECURITY.md#untrusted-models for more details). In a future release, the default value for `weights_only` will be flipped to `True`. This limits the functions that could be executed during unpickling. Arbitrary objects will no longer be allowed to be loaded via this mode unless they are explicitly allowlisted by the user via `torch.serialization.add_safe_globals`. We recommend you start setting `weights_only=True` for any use case where you don't have full control of the loaded file. Please open an issue on GitHub for any issues related to this experimental feature.
data = torch.load(data_path)
100%|██████████| 136/136 [00:01<00:00, 94.34it/s]
100%|██████████| 137/137 [00:01<00:00, 94.55it/s]
Latent embeddings generated¶
ls "../data/sim_sample/embedding"
sim_sample_data_tile0input_tx_id.csv sim_sample_data_tile1input_tx_id.csv
sim_sample_data_tile0.npy sim_sample_data_tile1.npy
Paried with the .npy (storing the latent embedding for input transcripts), there is the *input_tx_id.csv file. It stores the row index id for input transcripts in the “../data/sim_sample/raw/sim_sample.csv” to ensure the transcript meta information matches with the latent embedding.
Cluster the transcripts using the embedding matrix¶
SpatialRNA provides a helper function to run KMeans clustering (from sklearn package) on the embedding npy files for all tiles. We also show how to do fast (GPU-accelerated) clustering using PyCave library. See Case study repo ./code/run_gmm.py.
from spatialrna import run_kmeans
run_kmeans.run_kmeans(root="../data/",
sample_name_list =["sim_sample"],
downsample_to=None, # when the number of input transcripts are huge
downsample_seed=1024,
n_clusters=10,
split_file_per_sample=True,
verbose=False)
['../data/sim_sample/embedding/sim_sample_data_tile0.npy', '../data/sim_sample/embedding/sim_sample_data_tile1.npy']
['../data/sim_sample/embedding/sim_sample_data_tile0input_tx_id.csv', '../data/sim_sample/embedding/sim_sample_data_tile1input_tx_id.csv']
The clustering labels are saved in
ls ../data/sim_sample/clusters/
sim_sample.10clusters.csv
Plot results¶
from spatialrna import viz
import importlib
importlib.reload(viz)
We provided two visualisation function in SpatialRNA to demonstrate example ways of visualising the transcript clusters. We first gather the transcript meta information and the cluster labels for the sample we would like to plot using the get_tx_plot_df function.
tx_meta_with_clusters = viz.get_tx_plot_df(
root="../data/",
sample_name="sim_sample",
n_clusters=10)
tx_meta_with_clusters
?viz.plot_pixel
Signature:
viz.plot_pixel(
tx_meta,
pixel_size: float = 10,
min_points: int = 5,
x='X',
y='Y',
cluster_labels='cluster_labels',
figsize=(8, 8),
cmap=None,
join_method='avg',
background_color='white',
output_path=None,
**kwargs,
)
Docstring:
Pixelate points and color each pixel by either:
- average RGB of all points ("avg"), or
- major label color ("major").
Pixels with < min_points are set to background.
File: /mnt/beegfs/mccarthy/general/backed_up/rlyu/Projects/spatialrna_dev0.2/spatialrna/spatialrna/viz.py
Type: functionWe can generate pixel images and specify the size of the pixel:
colors = ['#e6194b', '#3cb44b', '#ffe119', '#4363d8', '#f58231',
'#911eb4', '#46f0f0', '#f032e6', '#bcf60c', '#fabebe', '#008080','#e6beff',
'#9a6324', '#fffac8', '#800000', '#aaffc3', '#808000', '#ffd8b1',
'#000075', '#808080', '#ffffff', '#000000']
#len(colors)
customise_cmap_k15 = dict(zip([x for x in range(10)],colors))
import matplotlib.pyplot as plt
plt.rcParams['figure.facecolor'] = 'black'
plt.style.use('dark_background')
p_fig,ax = viz.plot_pixel(tx_meta = tx_meta_with_clusters,
pixel_size=0.5,
min_points=1,
cmap=customise_cmap_k15,
background_color="black",
join_method="avg",
dpi=300,figsize=(8,8))
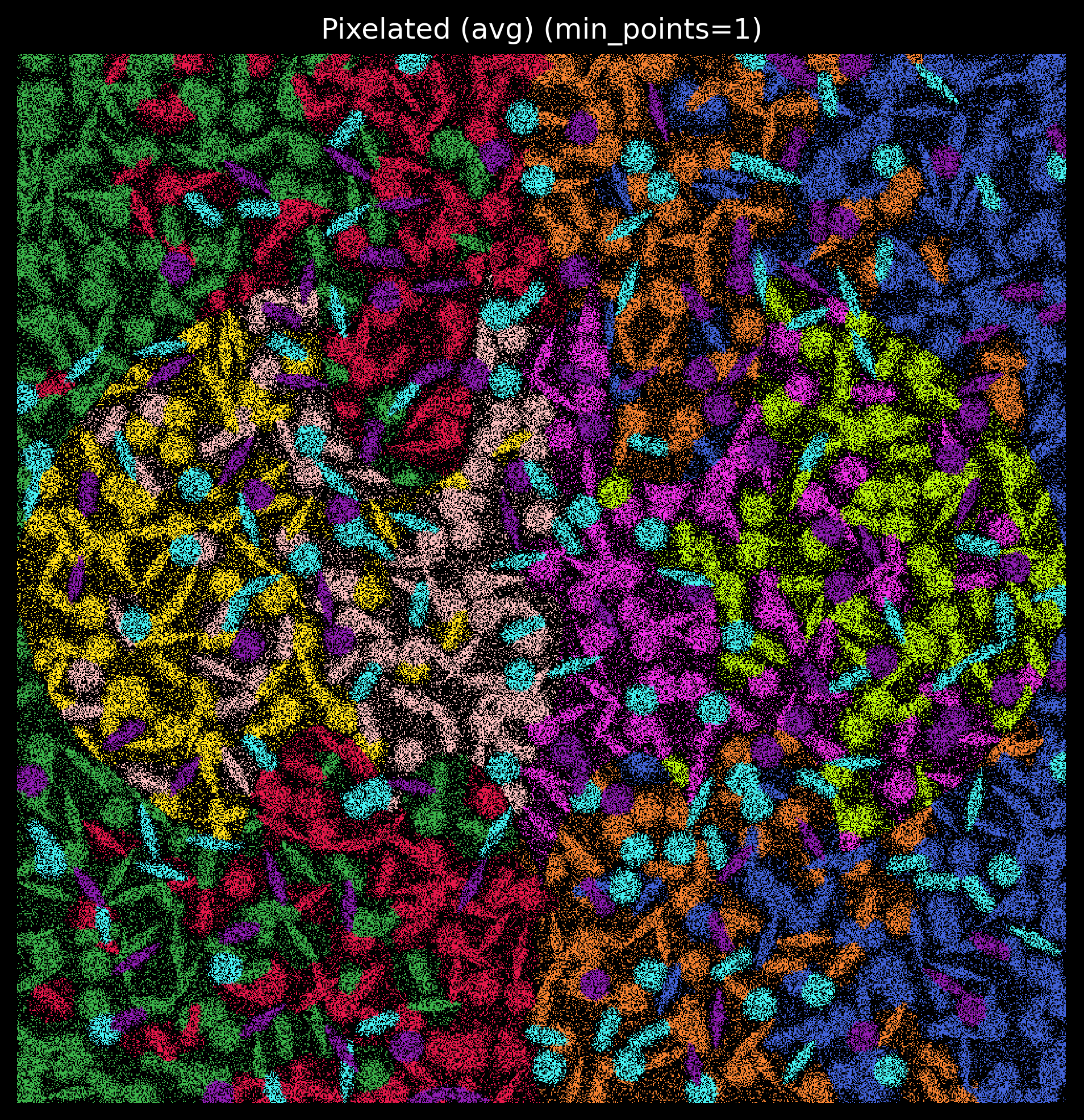
Match kmeans labels to simulated cell type labels¶
Since we have the ground-truth cell type labels, we can match the cluster ids to each cell type label, and quantify how well the clustering results capture the cell types.
groundtruth_label = pd.read_csv("../data/pixel_label.uniq.tsv.gz",sep="\t")
groundtruth_label.shape
(557521, 5)tx_meta_with_clusters.shape
(557521, 8)groundtruth_label["cluster_labels"] = tx_meta_with_clusters["cluster_labels"]
contingency_table = pd.crosstab(groundtruth_label.cell_label,groundtruth_label["cluster_labels"])
contingency_table
long_format = contingency_table.stack().reset_index()
long_format.columns = ['cell_labels', 'kmeans', 'Count']
long_format
long_format['Proportion'] = long_format.groupby('cell_labels')['Count'].transform(lambda x: x / x.sum())
long_format
sorted_pairs = long_format.sort_values(by='Proportion', ascending=False)
match_pair = long_format.iloc[long_format.groupby('cell_labels')['Proportion'].idxmax()]
The kmeans cluster labels are mapped to the groundtruth labels, for example kmeans cluster 5 corresponds to cell type kupffer_cell, and cluster 6 correspons to celltype granulocytes.
match_pair
match_pair_dict = dict(zip(match_pair.kmeans.astype(str) ,match_pair.cell_labels))
match_pair_dict
{'5': 'Kupffer_cell',
'9': 'cardiac_muscle_cell',
'8': 'cell_of_skeletal_muscle',
'4': 'endothelial_cell',
'0': 'epithelial_cell_of_proximal_tubule',
'1': 'fibroblast',
'6': 'granulocyte',
'2': 'hepatocyte',
'7': 'immature_NK_T_cell',
'3': 'keratinocyte'}kmeans_cell_label = [match_pair_dict[str(x)] for x in groundtruth_label["cluster_labels"]]
#Using the same color coding as before
## specify color maps
ground_truth_rgb = pd.read_csv("../data/model.rgb.tsv", sep="\t")
ground_truth_rgb
c_array = np.array(ground_truth_rgb[["R","G","B"]])
color_m = dict(zip(ground_truth_rgb.cell_label,range(0,10)))
color_m
{'Kupffer_cell': 0,
'granulocyte': 1,
'fibroblast': 2,
'epithelial_cell_of_proximal_tubule': 3,
'endothelial_cell': 4,
'keratinocyte': 5,
'hepatocyte': 6,
'cardiac_muscle_cell': 7,
'immature_NK_T_cell': 8,
'cell_of_skeletal_muscle': 9}plt.figure(figsize=(8,8))
plt.rcParams['figure.facecolor'] = 'black'
plt.style.use('dark_background')
plt.scatter(
x = groundtruth_label.X,
y = groundtruth_label.Y,
s=0.01,
c=[c_array[color_m[x]] for x in kmeans_cell_label],alpha=1)
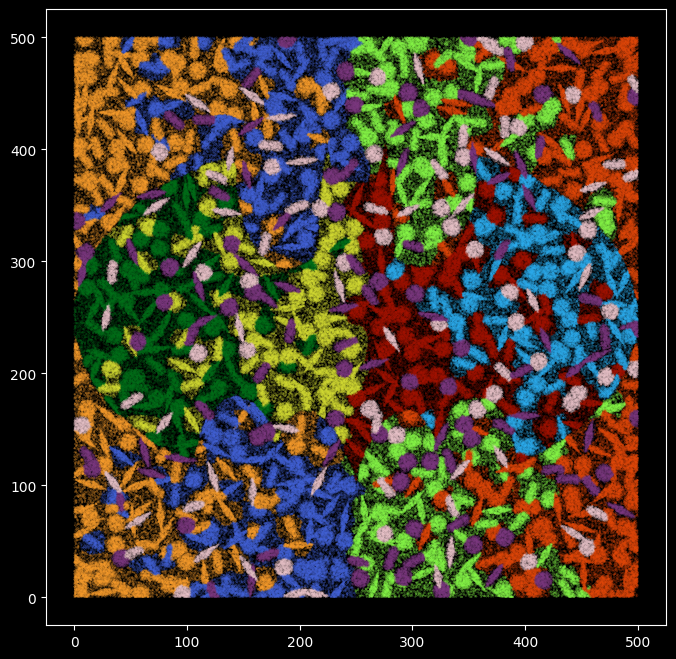
Quantify the consistency between kmeans labels and groundtruth cell type labels¶
np.array([kmeans_cell_label == groundtruth_label.cell_label]).sum()/groundtruth_label.shape[0]
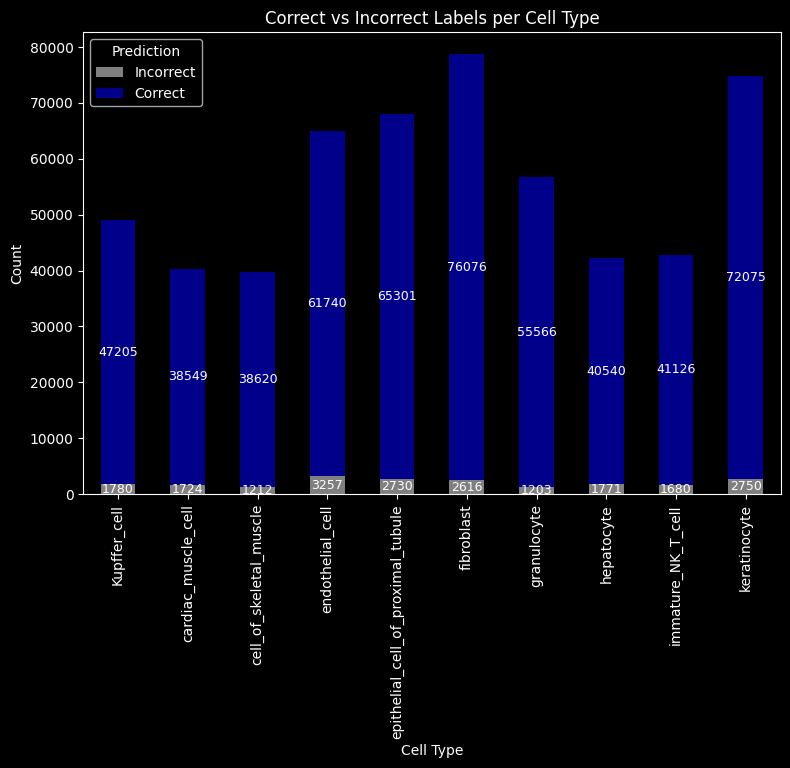
np.float64(0.9628300996733755)96% of the transcripts were assigned with the correct cell type labels.
import numpy as np
import pandas as pd
import matplotlib.pyplot as plt
# assume these exist
# kmeans_cell_label: np.array or pd.Series
# groundtruth_label.cell_label: np.array or pd.Series
pred = np.array(kmeans_cell_label)
true = np.array(groundtruth_label.cell_label)
# put into DataFrame for grouping
df = pd.DataFrame({"true": true, "pred": pred})
# compute correct/incorrect per cell type
df["correct"] = df["true"] == df["pred"]
summary = df.groupby("true")["correct"].value_counts().unstack(fill_value=0)
# rename columns for clarity
summary = summary.rename(columns={True: "Correct", False: "Incorrect"})
# plot
ax = summary.plot(kind="bar", stacked=True, figsize=(9, 6),
color={"Correct": "darkblue", "Incorrect": "grey"})
ax.set_ylabel("Count")
ax.set_xlabel("Cell Type")
ax.set_title("Correct vs Incorrect Labels per Cell Type")
ax.legend(title="Prediction")
# annotate bars
for p in ax.patches:
height = p.get_height()
if height > 0:
ax.annotate(f"{int(height)}",
(p.get_x() + p.get_width() / 2, p.get_y() + height / 2),
ha="center", va="center", fontsize=9, color="white")
plt.show()
groundtruth_label["kmeans"] = kmeans_cell_label
#groundtruth_label.to_csv("../data/kmeans_out_dmax3.csv")
Highlight the transcripts with inconsistent cell type labels¶
We now visualize the inconsistent transcripts, and we can see that those are likely to be from the outer layer (background) of a cell.
from itertools import compress
wrong_p = kmeans_cell_label != groundtruth_label.cell_label
pd.DataFrame(list(compress(kmeans_cell_label,wrong_p))).astype("category").value_counts()
0
granulocyte 7259
Kupffer_cell 4933
fibroblast 1545
endothelial_cell 1477
epithelial_cell_of_proximal_tubule 1294
cardiac_muscle_cell 1117
keratinocyte 1117
immature_NK_T_cell 708
hepatocyte 667
cell_of_skeletal_muscle 606
Name: count, dtype: int64Highlight in-consistent transcripts¶
Transcripts that have been correctly classified are colored with grey, the inconsistent transcripts are highlighted and colored by groundtruth transcript labels.
# wrong_p = kmeans_cell_label != true_label.cell_label
plt.figure(figsize=(7,7))
plt.rcParams['figure.facecolor'] = 'black'
plt.scatter(y=groundtruth_label.Y[wrong_p],x=groundtruth_label.X[wrong_p],s=0.1,
c=[c_array[color_m[x]] for x in list(compress(kmeans_cell_label,wrong_p))],alpha=1)
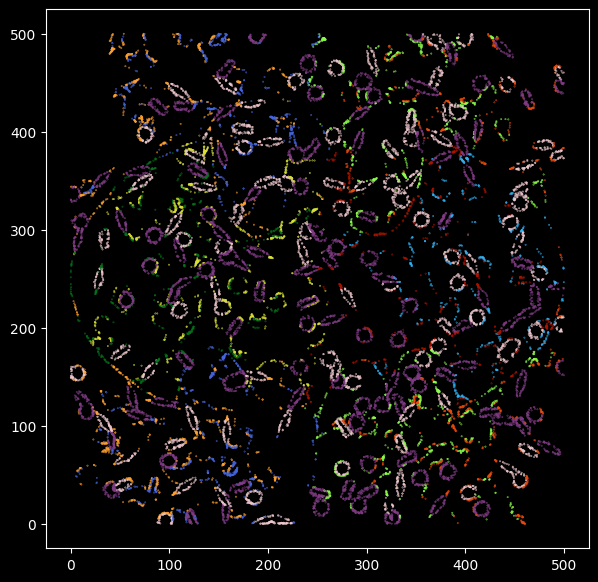
boolean_vector = pd.Series(wrong_p)
# Map True to 0.5 and False to 1
numeric_vector = boolean_vector.map({True: 1, False: 0.1})
c_map = [c_array[color_m[x]] for x in groundtruth_label.cell_label]
#pd.Series(wrong_p)
cmap_m = np.stack(c_map)
cmap_m[~wrong_p,:] = 0.60
cmap_m
array([[0.6, 0.6, 0.6],
[0.6, 0.6, 0.6],
[0.6, 0.6, 0.6],
...,
[0.6, 0.6, 0.6],
[0.6, 0.6, 0.6],
[0.6, 0.6, 0.6]], shape=(557521, 3))plt.rcParams['figure.facecolor'] = 'black'
plt.rcParams['figure.dpi'] = '150'
plt.figure(figsize=(5,5))
# wrong_p = kmeans_cell_label != true_label.cell_label
plt.scatter(x=groundtruth_label.X,y=groundtruth_label.Y,s=0.1,
c=cmap_m, alpha=numeric_vector)
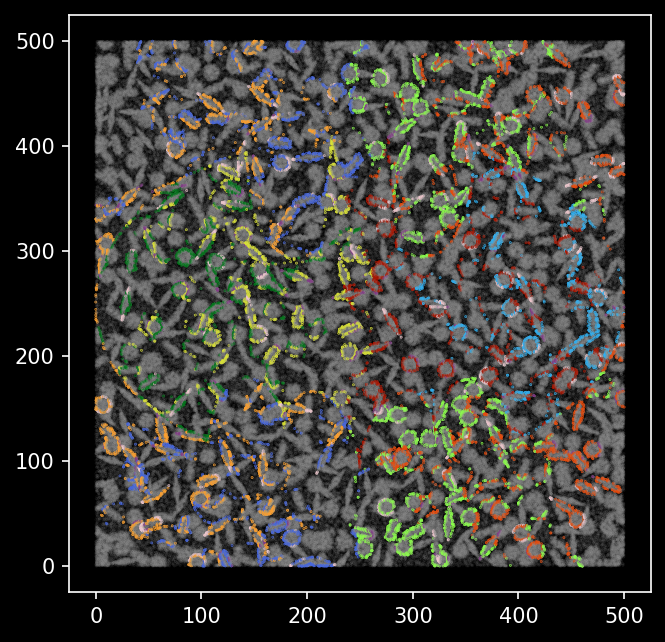
We see the transcripts that were classified differently from the groundtruth cell type labels are mostly outlining the boundaries of cells.
ARI score¶
We can calculate the ARI score between our clusters derived based on latent embeddings and the ground-truth cell labels.
from sklearn.metrics import adjusted_rand_score
ari_score = adjusted_rand_score(groundtruth_label.cell_label, groundtruth_label.kmeans)
ari_score
0.9198942906061998Put together¶
plt.style.use('dark_background')
plt.rcParams['figure.facecolor'] = 'black'
plt.rcParams['figure.dpi'] = '100'
fig,axes = plt.subplots(1,3,figsize=(12,4),sharey=True)
fig.tight_layout(h_pad=0.5)
axes[0].scatter(y = groundtruth_label.X,x = groundtruth_label.Y, s=0.01, c=[c_array[color_m[x]] for x in groundtruth_label.cell_label],alpha=1)
axes[0].set_title('Ground-truth labels')
axes[1].scatter(y=groundtruth_label.X,x=groundtruth_label.Y,s=0.01,c=[c_array[color_m[x]] for x in groundtruth_label.kmeans],alpha=1)
axes[1].set_title(f'Cluster labels from GNN ARI: {ari_score:.3f}')
axes[2].scatter(y=groundtruth_label.X,x=groundtruth_label.Y,s=0.01,c=cmap_m,alpha=numeric_vector)
axes[2].set_title('Transcripts with inconsistent prediction labels from groundtruth')
axes[0].axis('off')
axes[1].axis('off')
axes[2].axis('off')
(np.float64(-25.0), np.float64(525.0), np.float64(-25.0), np.float64(525.0))